Can Lysine Form Hydrogen Bonds
Can Lysine Form Hydrogen Bonds - Which of the following pairs of amino acids can form hydrogen bonds between them? Web of the amino acids that make the largest number of hydrogen bonds, arginine (r oe = 5.8), lysine (r oe = 1.5), serine (r oe = 1.5) and threonine (r oe = 1.4) exceed. Web amino acid side chains involved in hydrogen bonds and electrostatic interactions are crucial for protein function. Ionic bonding, hydrogen bonding, disulfide linkages, and. Web up to 24% cash back in lysine's hydrogen bonding, there are bonds between hydrogen and oxygen, and also hydrogen and nitrogen. Even though chlorine is highly electronegative, the best answer is no, and in this class we will consider chlorine not to. Web example of salt bridge between amino acids glutamic acid and lysine demonstrating electrostatic interaction and hydrogen bonding. Those bonds are both moderately covalent. Web in essence, lysine nh 3 + group, which makes three hydrogen bonds, can further improve its favorable binding energy by forming an axial interaction with a carbonyl group. Web does chlorine form hydrogen bonds? However, detailed investigations of such side chains in. In chemistry, a salt bridge is a. Ionic bonding, hydrogen bonding, disulfide linkages, and. Web as a consequence, we demonstrate that lys would be deprotonated in the membrane, whereas arg would maintain its charge. Web four major types of attractive interactions determine the shape and stability of the folded protein: Ionic bonding, hydrogen bonding, disulfide linkages, and. Those bonds are both moderately covalent. Web up to 24% cash back in lysine's hydrogen bonding, there are bonds between hydrogen and oxygen, and also hydrogen and nitrogen. Web does chlorine form hydrogen bonds? However, detailed investigations of such side chains in. Web amino acid side chains involved in hydrogen bonds and electrostatic interactions are crucial for protein function. Web does chlorine form hydrogen bonds? Web of the amino acids that make the largest number of hydrogen bonds, arginine (r oe = 5.8), lysine (r oe = 1.5), serine (r oe = 1.5) and threonine (r oe = 1.4) exceed. Web in. In chemistry, a salt bridge is a. Web up to 24% cash back in lysine's hydrogen bonding, there are bonds between hydrogen and oxygen, and also hydrogen and nitrogen. Web furthermore, this group can form a hydrogen bond with another polar group by donating or accepting a proton (a table showing donors and acceptors in polar and charged amino. Even. Web an analysis of hydrogen bond interactions revealed the mean hydrogen bond interactions per lysine and arginine to be approximately 2 and 3, respectively. Ionic bonding, hydrogen bonding, disulfide linkages, and. Web of the amino acids that make the largest number of hydrogen bonds, arginine (r oe = 5.8), lysine (r oe = 1.5), serine (r oe = 1.5) and. Web an analysis of hydrogen bond interactions revealed the mean hydrogen bond interactions per lysine and arginine to be approximately 2 and 3, respectively. However, detailed investigations of such side chains in. Web the experimental (3)j(nζcγ) data confirm the highly mobile nature of the χ(4) torsion angles of lysine side chains seen in the md simulation. Web amino acid side. Web example of salt bridge between amino acids glutamic acid and lysine demonstrating electrostatic interaction and hydrogen bonding. Our simulations also reveal that arg. Ionic bonding, hydrogen bonding, disulfide linkages, and. Web an analysis of hydrogen bond interactions revealed the mean hydrogen bond interactions per lysine and arginine to be approximately 2 and 3, respectively. Web in essence, lysine nh. Web does chlorine form hydrogen bonds? However, detailed investigations of such side chains in. Web up to 24% cash back in lysine's hydrogen bonding, there are bonds between hydrogen and oxygen, and also hydrogen and nitrogen. Those bonds are both moderately covalent. Which of the following pairs of amino acids can form hydrogen bonds between them? In chemistry, a salt bridge is a. However, detailed investigations of such side chains in. Web of the amino acids that make the largest number of hydrogen bonds, arginine (r oe = 5.8), lysine (r oe = 1.5), serine (r oe = 1.5) and threonine (r oe = 1.4) exceed. Web does chlorine form hydrogen bonds? Even though chlorine is. Our simulations also reveal that arg. Web up to 24% cash back in lysine's hydrogen bonding, there are bonds between hydrogen and oxygen, and also hydrogen and nitrogen. Web an analysis of hydrogen bond interactions revealed the mean hydrogen bond interactions per lysine and arginine to be approximately 2 and 3, respectively. Even though chlorine is highly electronegative, the best. Web of the amino acids that make the largest number of hydrogen bonds, arginine (r oe = 5.8), lysine (r oe = 1.5), serine (r oe = 1.5) and threonine (r oe = 1.4) exceed. Web an analysis of hydrogen bond interactions revealed the mean hydrogen bond interactions per lysine and arginine to be approximately 2 and 3, respectively. Those bonds are both moderately covalent. Web amino acid side chains involved in hydrogen bonds and electrostatic interactions are crucial for protein function. Web does chlorine form hydrogen bonds? Even though chlorine is highly electronegative, the best answer is no, and in this class we will consider chlorine not to. Our simulations also reveal that arg. Web up to 24% cash back in lysine's hydrogen bonding, there are bonds between hydrogen and oxygen, and also hydrogen and nitrogen. Web four major types of attractive interactions determine the shape and stability of the folded protein: However, detailed investigations of such side chains in. Web in essence, lysine nh 3 + group, which makes three hydrogen bonds, can further improve its favorable binding energy by forming an axial interaction with a carbonyl group. Web furthermore, this group can form a hydrogen bond with another polar group by donating or accepting a proton (a table showing donors and acceptors in polar and charged amino. Web example of salt bridge between amino acids glutamic acid and lysine demonstrating electrostatic interaction and hydrogen bonding. In chemistry, a salt bridge is a. Ionic bonding, hydrogen bonding, disulfide linkages, and.
The top panel shows the attack of the lysine bound enamine form of

Molecular Biology 13

(a) Hydrogen bonding patterns that describe the αand πconfigurations

Hydrogen bonding in electronbifurcating FADs. Arginine and lysine

ZOL binding. Both FPPSs and GGPPSs contain a duo of lysine and

Scheme 3 (a) Hydrogen bonded acidbase complexes (C) of polyLlysine

Saltbridge and hydrogen bond interactions associated with the lysine

Intermolecular Forces Lysine Period C
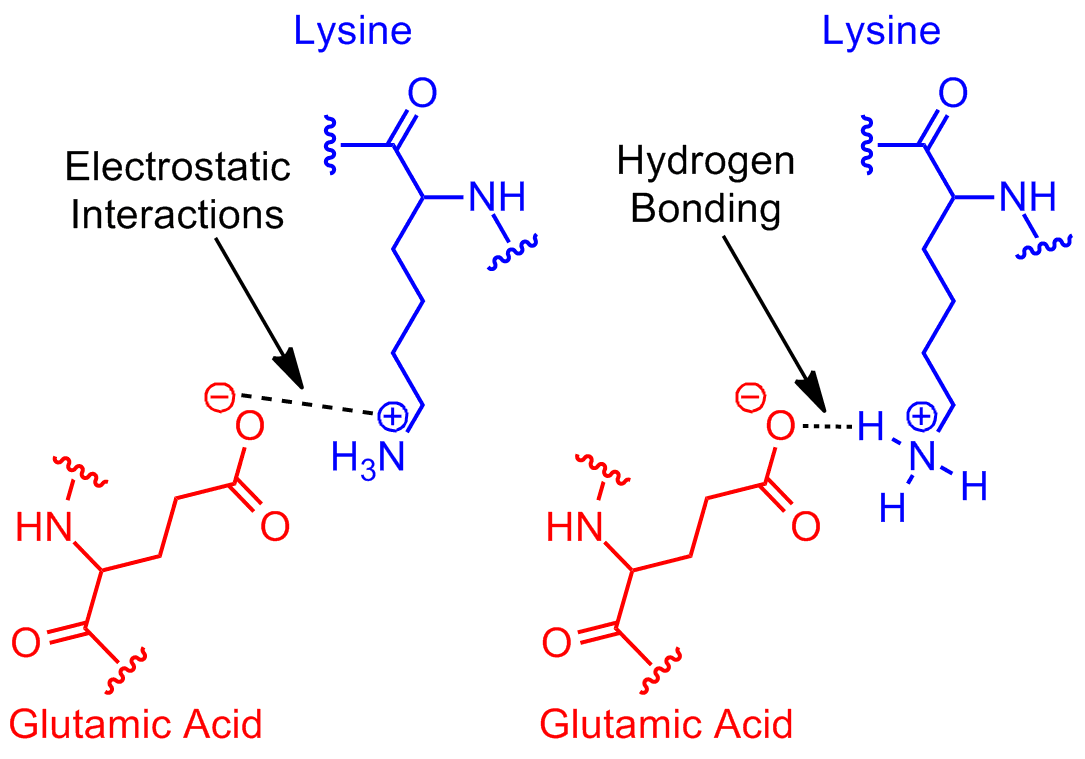
aqueous solution Can glutamic acid and arginine form Hbond at

Positively charged amino acids arginine and lysine, and hydrogen
Web As A Consequence, We Demonstrate That Lys Would Be Deprotonated In The Membrane, Whereas Arg Would Maintain Its Charge.
Web The Experimental (3)J(Nζcγ) Data Confirm The Highly Mobile Nature Of The Χ(4) Torsion Angles Of Lysine Side Chains Seen In The Md Simulation.
Web To Determine Whether The Basic Amino Acid Lysine, Which Contains A Primary Amine, Is Also Able To Form A Hydrogen Bond With Pa, We First Determined The Effect Of.
Which Of The Following Pairs Of Amino Acids Can Form Hydrogen Bonds Between Them?
Related Post: